Q466-04
Hex-1-ene gas, C
6H
12, burns in oxygen to produce carbon dioxide and water
vapour. Write an equation to represent this reaction.
| show |
C6H12(g) + 9O2(g)  6CO2(g) + 6H2O(g)
6CO2(g) + 6H2O(g) |
|
|
Use the data below to calculate the values of ΔHc

and ΔSc

for the combustion of hex-1-ene.
|
Substance
|
O2(g) |
C6H12(g) |
CO2(g) |
H2O(g) |
Standard enthalpy of formation, ΔHf 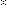 /kJmol -1 |
0.0 |
-43 |
-394 |
-242 |
Entropy, S 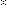 /JK -1
mol -1 |
205 |
385 |
214 |
189 |
| show
|
|
Reactants enthalpy of formation
1 x C6H12(g) = -43 kJ
|
Products enthalpy of formation
6 x CO2(g) = 6 x -394 = -2364 kJ
6 x H2O(g) = 6 x -242 = -1452 kJ
Total = -3816 kJ
|
|
The reactants are 'unformed' so change the sign of
the reactants and add it to the products:
Reaction enthalpy = Products ΔHf - ΔReactants
Hf = -3816 - (-43) = -3773
kJ
|
|
Reactants entropy
1 x C6H12(g) = 1 x 385 = 385
JK-1
9 x O2(g) = 9 x 205 = 1845 JK-1
Total = 2230 JK-1
|
Products entropy
6 x CO2(g) = 6 x 214 = 1284 JK-1
6 x H2O(g) = 6 x 189 = 1134 JK-1
Total = 2418 JK-1
|
|
There is an increase in entropy by 2418 - 2230 = +188
JK-1
ΔSc = +188 JK-1
= +188 JK-1
|
|
|
Calculate the standard free energy change for the combustion of hex-1-ene.
| show
|
| Using the Gibbs free energy equation:
At 298 K: ΔG = -3773 - 298(0.188) = -3773 - 56 = -3829
kJ
|
|
State and explain whether or not the combustion of hex-1-ene is spontaneous
at 25ºC
| show |
| The final value for Gibbs free energy is negative at 298K,
so the reaction is spontaneous. |
|
Study
question 1 Study
question 2 Study
question 3 Study
question 5