|
The students are required to "perform lab experiments which could include single replacement reactions in aqueous solutions". This is sufficiently vague to allow for several types of experiment: |
Precipitation reactions could be carried out to investigate water of crystallisation.
The water of crystallisation in iron(II) ammonium sulfate - procedure
Hydrated iron(II) ammonium sulfate has the formula Fe(NH4)2(SO4)2.xH2O.
Accurately weigh approximately 5 g of iron(II) ammonium sulfate and dissolve in 100 ml of water. The solution is then made up to 250 ml in a volumetric flask.
Transfer a 25 ml aliquot to a conical flask and add 25 ml of 1 mol dm-3 (excess) barium chloride solution.
Filter the precipitation into a weighed filter paper, wash with a little water and dry in an oven.
The dry precipitate and filter paper are then weighed.
The procedure was repeated using fresh 25 ml aliquots of the iron(II) ammonium sulfate solution.
Data recording
All data should be recorded with associated uncertainty.
Mr BaSO4 = 233.3
Mass of hydrated iron(II) ammonium sulfate = 5.13 g ± 0.01
Mass of filter paper = 0.52 g ± 0.01
Mass of filter paper + dry precipitate = 6.61 g ± 0.01
Observations: The barium chloride formed a dense white precipitate on addition to the iron ammonium sulfate solution.
Data analysis
Mass of BaSO4 precipitate = 6.61 - 0.52 = 6.09 g
Moles of BaSO4 precipitate = 6.09/233.3 = 0.0261 mol
Hence, moles of sulfate ions = 0.0261 mol
Fe(NH4)2(SO4)2.xH2O
+ (aq) ![]() Fe2+(aq)
+ 2NH4+(aq) + 2SO42-(aq) + xH2O(l)
Fe2+(aq)
+ 2NH4+(aq) + 2SO42-(aq) + xH2O(l)
Ba2+(aq) + SO42-(aq)
![]() BaSO4(s)
BaSO4(s)
Hence, moles of iron ammonium sulfate = 0.0261/2 = 0.0131 mol
Mass of Fe(NH4)2(SO4)2 = moles x Mr = 0.0131 x 284 = 3.71 g
Hence, mass of water of crystallisation = original mass - mass of iron ammonium sulfate = 5.13 - 3.71 = 1.42 g
Moles of water of crystallisation = 1.42/18 = 0.0789 mol
Hence, ratio of iron ammonium sulfate to water of crystallisation = 0.0131 : 0.0789 = 1 : 6.02
Hence, formula of hydrated iron(II) ammonium sulfate = Fe(NH4)2(SO4)2.6H2O
Errors and inaccuracies
Mass of hydrated iron ammonium sulfate = 5.13 ± 0.01 = 0.19 %
Mass of BaSO4 precipitate = 6.61 - 0.52 = 6.09 g ± 0.02 = 0.33 %
Volumetric solution = 250 ml ± 0.26 = 0.10 %
Pipette = 25 ml ± 0.15 = 0.6 %
Total percentage error = 1.22 %
As we are looking for integral values for the water of crystallisation this inaccuracy lies well within our range.
The nucleophilic substitution of halogenoalkanes could be investigated in terms of the rate of reaction.
Experimental - The reaction between sodium hydroxide and 2-bromopropane
NaOH(aq) + CH3CHBrCH3(aq)
![]() NaBr(aq) + CH3CHOHCH3(aq)
NaBr(aq) + CH3CHOHCH3(aq)
The products of this reaction can be readily determined. The concentration of the sodium hydroxide decreases as the reaction proceeds or the bromide ions formed can be determined by precipitation with silver ions (after neutralising the NaOH using dilute nitric acid).
Procedure
A dilute solution of 2-bromopropane is prepared by dissolving a known mass of the compound in a 50:50 ethanol water mixture and making up to the mark in a 250 ml volumetric flask.
A 0.1 mol dm-3 solution of sodium hydroxide is prepared by weighing out about 1.0g of the compound and dissolving it in about 100 ml of water, and then making up to the mark in a 250 ml volumetric flask.
A 100 ml aliquot of the sodium hydroxide solution is added to a 100 ml aliquot of the 2-bromopropane solution and the timer started.
After an interval a 25 ml portion is removed from the reaction mixture and pipetted into 25 ml of 0.2 mol dm-3 nitric acid.
The excess acid can then be titrated against a standard sodium hydroxide solution.
NaOH(aq) + HNO3(aq) ![]() NaNO3(aq) + H2O(l)
NaNO3(aq) + H2O(l)
The extraction and determination is then repeated at regular intervals.
Data analysis
The moles of sodium hydroxide remaining in the reaction mixture can be found from the initial moles of nitric acid - moles acid at titration.
Moles of sodium hydroxide reacted = initial moles of sodium hydroxide - moles remaining.
The concentration of sodium hydroxide in the reaction mixture can then be determined (moles/volume).
A graphical plot of sodium hydroxide concentration against time can then be used to find the rate of reaction from the gradient of the tangent to the curve.
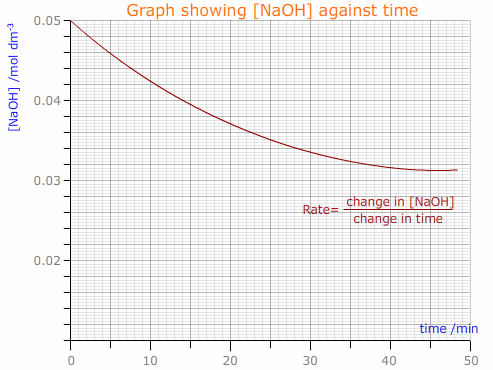
As the data is used to determine the rate of reaction, the experiment can be adapted to investigate the effect of other variables on the reaction. It would be possible to change the solvent used (ethanol/water in this case) or the temperature, etc.
Halogens are oxidising agents and the higher members of group 17 oxidise the ions of the lower members. This was covered in oxidation and reduction - section 9.31.
These reactions can be used to determine the concentration of halogens in dilute solutions.
Experiment - determination of chlorine in bleach
Bleach is a dilute solution of sodium chlorate(I), NaClO, a compound that easily releases chlorine in the presence of acids:
NaClO + 2HCl ![]() NaCl + Cl2 + H2O
NaCl + Cl2 + H2O
The reaction is an equilibrium and any compound that reacts with chlorine removes it from the equilibrium and pushes the system into making more, until it is all exhausted (used up). This can be used to determine the concentration of the sodium chlorate(I)
Procedure
A 25 ml sample of household bleach is pipetted into a volumetric flask and the volume made up to the 250 ml mark using distilled (deionised) water.
A 25 ml aliquot is extracted from this solution into a conical flask and 25 ml of a 0.2 mol dm-3 potassium iodide (KI) solution added.
The liberated iodine is then titrated against a standard 0.02 mol dm-3 solution of sodium thiosulfate until the yellow colour is nearly discharged.
5 ml of freshly prepared starch solution is added and the titration is continued until the blue-black starch iodide colour is completely discharged (colourless).
The titration is repeated until concordant results are obtained.
Data recording
As in previous titrations all measurements and readings are recorded with associated uncertainties. This includes qualitative data (observations).
Data analysis
The reaction that occurs between chlorine and potassium iodide produces iodine:
1 Cl2
+ KI ![]() 2KCl + I2
2KCl + I2
So when excess potassium iodide is used all of the chlorine is reduced and makes a stoichiometric amount of iodine.
The liberated iodine reacts with thiosulfate ions:
2 I2
+ 2S2O32- ![]() S4O62- + 2I-
S4O62- + 2I-
Measuring the volume of sodium thiosulfate needed gives the moles of thiosulfate ions = molarity x volume.
From equation 2 moles of thiosulfate/2 = moles of iodine.
From equation 1 moles of iodine formed = moles of chlorine reacted.
From the sodium chlorate(I) equilibrium moles of chlorine = moles of sodium chlorate(I).
If we have the moles of sodium chlorate(I) in a 25 ml aliquot, we can multiply by 10 to obtain the moles of sodium chlorate(I) in 250 ml.
This is equal to the moles of sodium chlorate(I) in the original 25 ml sample extracted from the commercial bleach bottle.
Hence, the concentration of sodium chlorate(I) in bleach =moles of sodium chlorate(I)/0.025 mol dm-3.
Displacement of metal ions by more reactive metals is a standard thermochemistry experiment used to determine the enthalpy change of reaction by back-extrapolation. This has been covered in "Energetics" - Section 4.17.
