|
The ideal gas law gives the relationship between the number of moles of a gas and the pressure, volume and temperature. PV = nRT This can be used to find the moles of volatile substance and, if the mass is known, the relative molecular mass of the substance. |
The gas syringe
A gas syringe is a piece of equipment that can be used in an oven to measure the volume of a volatile substance at different temperatures.
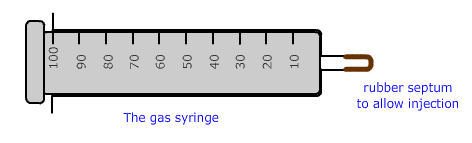
Part 1 - preparing the syringe
Accurately weigh the gas syringe
Introduce a small portion of the liquid through the rubber septum using a microsyringe and needle
Reweigh the gas syringe and compound
Part 2 - the oven
Place the gas syringe in a suitably calibrated oven and allow to reach the desired temperature
Read the volume obtained on the syringe.
Repeat the procedure to reduce random error
Ensure that all of the data is recorded with suitable units and inaccuracies.
The following shows example raw data and analysis.
Raw data
Mass of gas syringe: 58.63 g ± 0.01
Mass of gas syringe + compound: 58.94 g ± 0.01
Temperature of oven: 373K ± 0.5
Pressure: 100.5 kPa ± 0.5
Volume obtained: 57.0 ± 0.5 cm3 = 0.057 dm3
Data analysis
Mass of compound used = 58.94 - 58.63 = 0.31 g ± 0.02
Using the ideal gas equation: PV = nRT therefore n = PV/RT
number of moles = (100.5 x 0.057) / (8.314 x 373) = 5.7285/3101.122 = 0.001847 mol
As Mr = mass/mol = 0.31 / 0.001847 = 167.8
Treatment of errors and inaccuracies
Take a percentage error of each of the absolute inaccuracies and sum them:
Gas volume inaccuracy = 100 x 0.5/57 = 0.88%
Temperature: 100 x 0.5/373 = 0.13%
Pressure: 100 x 0.5/100.5 = 0.5%
Mass: 100 x 0.02/0.31 = 6.5%
Accumulated percentage error = 8.01%
This value can then be applied to the answer obtained
Relative mass of gas = 167.8 ± 13.4
