|
Phases are physically distinct states of matter. This section looks at the behaviour of phases and changes between them. |
|
Phase equilibrium
States of matter may be described as phases. Phases are physically distinct states with an obvious boundary between them.
For example two immiscible liquids are both in the same state (they are liquid), but different phases. There is a distinct boundary between them.
Equilibria can exist between two phases, whether by change of state, or by transport between the phases.
|
Example: Iodine is soluble in both tetrachloromethane and water. In tetrachloromethane it forms a purple solution and in water it is less soluble and forms a brown solution. If iodine is shaken with a mixture of water and tetrachloromethane and allowed to settle, an equilibrium is established in which some of the iodine is dissolved in the aqueous layer and some in the tetrachloromethane layer. Iodine continues to pass from one layer to the other and vice versa across the boundary. |
Changes of state can also be described as phase changes. A phase equilibrium is a reversible process involving phase change in which a dynamic equilibrium has been established.
| liquid water and water vapour |
These forward and reverse processes can be represented:
liquid water 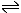 water
vapour water
vapour |
Solid - liquid phases may also establish an equilibrium, for example, in a mixture of ice and water at 0ºC.
Position of equilibrium
Once an equilibrium is established, the relative amounts of all of the components don't change.
An equilibrium can be thought of as having a balancing point - the position of equilibrium, given by the relative amounts of left hand side and right hand side.
This allows us to define a constant that relates to the amounts of the components. A phase equilibrium can be defined by the ratio of solute concentration found in the two phases.
|
Example: In the phase equilibrium of iodine
in water and tetrachloromethane, the equilibrium constant for the process:
I2(CCl4) K = [I2(H2O)]/[I2(CCl4)] |
If the system is changed by adding or removing matter or energy, then if this causes more of the right hand side components to be formed, we say that the position of equilibrium has shifted to the right.
Likewise, if more of the left hand side is formed by some change in conditions, then we say that the position of equilibrium has shifted to the left.
NOTE If the position of equilibrium is disturbed by adding (or subtracting) one or more of the components, the system responds in such a way as to restore the value of the equilibrium constant.
Once a situation of dynamic equilibrium is attained the rate of the forward process is equal to the rate of the reverse process and concentration of reactants and products remains constant.
This constant value is called the equilibrium constant; it is assigned the letter k.
If the equilibrium constant is defined in terms of concentrations it is called kc, if it is defined in terms of partial pressures of gas it is designated the letter kp.
Both kc and kp are only dependent on temperature.
The equilibrium constant is given by the equation:
kc = [products]coeff/[reactants]coeff
Where:
- [products]coeff means the concentration of the products raised to the power of its coefficient in the balanced equation,
- [reactants]coeff means the concentration of the products raised to the power of its coefficient in the balanced equation
|
Example: The distribution coefficient for iodine between the two solvents water and tetrachloromethane is given by the equation and the equilibrium law expression: I2(aq) By the equilibrium law: k = [I2(CCl4)]/[I2(aq)]
At 298K this has a value of about 650 (dimentionless) |
The effect of changing the equation
The consequences of the coefficients being involved in the equilibrium law expression means that the equilibrium constant varies according to the reaction equation.
For example, the reversible decomposition of hydrogen iodide can be expressed using two different equations:
equation 1:
2HI(g) ![]() H2(g)
+ I2(g)
H2(g)
+ I2(g)
equation 2:
HI(g) ![]() ½H2(g)
+ ½I2(g)
½H2(g)
+ ½I2(g)
Each equation has a different kc value.

The equilibrium constant for HI decomposition (equation 1) at 500 °C is 5.8×10-3
The equilibrium constant for HI decomposition (equation 2) at 500 °C is 7.6 ×10-2
The reaction quotient, represented by the letter Q, measures the relative amounts of the products and the reactants of a reversible reaction at any particular time.
The reaction quotient is the value of the equilibrium law expression under non-equilibrium conditions.
If Q is not equal to the equilibrium constant then the reaction or process continues until the position of equilibrium is attained.
|
Example: Two immiscible solutions of iodine in water and tetrachloromethane are mixed together and the reaction quotient measured before shaking. If the distribution coefficient for the two solutions at the given temperature is 650 and the value of Q is measured to be 800, which way will iodine pass? The distribution coefficient is given by the expression: k = [I2(CCl4)]/[I2(aq)] = 650 But Q = 800, in other words the value for [I2(CCl4)] is relatively higher than it should be, so iodine will pass from the organic layer to the aqueous layer until the value of the distribution coefficient is attained. I2(CCl4)
|

